Periodic table chemistry sudafedrine12/4/2023 You just need to sit back and watch, and let the amazing natural power of your visual memory do its thing. This animated video course is the fastest way to memorize the periodic table because it uses best-practice visual memory techniques.Īll the mental images and association links described above have already been created, and transformed into engaging whiteboard animation videos. The weakness of this method compared to the Memory Palace is the amount of time it takes to create the (intentionally) bizarre and crazy story to link all the words (or chemical elements) together.īut all the work has already been done for you at How to Memorize the Periodic Table. The method used in the video above is called the Link and Story Method, and is based on the same principles of visualization and association. Google ‘memory palace’ or ‘world memory champion’ and you’ll discover they’re the fastest and most effective methods to memorize a deck of playing cards and a lot of other geeky things. These established memory techniques have been proven by over 50 years of academic research in fields like cognitive psychology. And when your bus begins talking with a ‘lithp’ (how people with a lisp pronounce ‘lisp’), you’ll be prompted to recall lithium. When you picture a large helium balloon tied to your front gate, you’ll remember helium. Because the chemical elements themselves can be difficult to visualize, you substitute them with an object that you will naturally associate or link to the element itself.įor example, ‘hydrogen’ sounds similar to ‘hydrant’, so when you visualize a hydrant sitting at your front door, you’ll be prompted to remember ‘hydrogen’. You might walk out your front door, through the front gate, and get on a bus.Īt each location you visualize an object that represents what you’re trying to remember. 
Think of a particular journey you take every day, and picture certain locations along the way.įor example, imagine leaving home in the morning and travelling to work or school. The foundation technique most memory experts use is the Method of Loci (or Memory Palace or Journey Method). Watch YouTube’s #1 “How to Memorize” video and you’ll probably amaze yourself with how easily you can remember and recall 15 random words in order, using one of these techniques. It’s incredibly simple but amazingly fast and effective. That’s a fancy way of saying they create mental pictures and link them together in their mind. Memory experts and world champion memory ‘athletes’ activate the enormous natural power of their visual memory by using visualization and association mnemonic techniques. That is, similar elements do not have similar atomic weights.You can memorize the periodic table in one night, simply by emulating best-practice memorization techniques and doing what memory experts do. This law states that when the elements are listed in order of increasing atomic weights, their properties vary periodically. If a list were made of all elements, we would find the sequence halogen, noble gas, alkali metal, and alkaline-earth metal several more times.ĭmitri Ivanovich Mendeleev proposed the periodic law behind his periodic table compiling. Exactly the same sequence is repeated eight elements later (Ne, Na, and Mg), but this time a halogen (F) precedes the noble gas. The second, third, and forth elements on the list (He, Li, and Be) are a noble gas, an alkali metal, and an alkaline-earth metal, respectively. Obtaining atomic weights, we haveĮlements which belong to families we have already discussed are indicated by shading around their symbols. You can see that such a relationship exist by listing symbols for the first dozen elements in order of increasing relative mass. Therefore it seems reasonable to expect some correlation between this microscopic property and macroscopic chemical behavior. This could account for the related chemical reactivities and analogous compounds of these elements.Īccording to Dalton’s atomic theory, different kinds of atoms may be distinguished by their relative masses (atomic weights). Atoms of sodium ought to be similar in some way to atoms of lithium, potassium, and the other alkali metals. The similarities among macroscopic properties within each of the chemical families lead one to expect microscopic similarities as well. 1950-1999 (+15 elements): Manhattan_Project and Particle physics issues, for atomic numbers 97 and above.  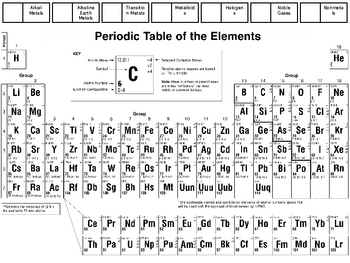
1900-1949 (+13 elements): impulse from the old quantum theory, the Refinements to the periodic table, and quantum mechanics.1850-1899 (+23 elements): the age of Classifying Elements received an impulse from the Spectrum analysis. 
1800-1849 (+22 elements): impulse from Scientific Revolution and Atomic theory and Industrial Revolution.Before 1800 (36 elements): discoveries during and before the Age of Enlightenment.\): Periodic Table showing when each element was discovered
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
